Disclaimer: This article is intended for industry professionals and provides general educational information only. It does not promote specific products or constitute medical advice.
Navigating a New Era in Global Trade
Trade tensions between China and the European Union are reshaping the medical device sector, creating a pivotal moment for UK businesses. Operating independently of EU policy constraints, post-Brexit, UK firms- including SMEs-can emerge as trusted leaders in a market projected to grow from £473 billion in 2024 to £501 billion in 2025 [Statista, 2024]. With strategic action, UK companies can capitalise on these shifts, navigating challenges such as regulatory complexities and competition from established players like the US and Japan.
A Fractured Playing Field: Turning Barriers into Bridges
Recent reciprocal restrictions have altered the competitive landscape, potentially reducing direct EU-China rivalry and opening niches for UK suppliers.
Key Restrictions at a Glance
- EU Measures: The European Commission has excluded Chinese companies from public procurement of medical devices exceeding £4.3 million (€5 million), with a 50% cap on Chinese-origin components in tenders.
- China’s Countermeasures: In response, China has restricted EU-origin medical devices in government contracts above £4.6 million (45 million yuan), enforcing a 50% local content requirement.
However, success demands expertise in China’s relationship-driven (guanxi) market and the EU’s bureaucratic procurement processes.
Why the UK is Poised to Lead
- Strategic Positioning
Free from the EU–China dispute, UK businesses can serve as pragmatic collaborators, appealing to stakeholders seeking stability. For example, UK MedTech firms have increasingly engaged in China through events like the UK-China Healthcare 2024 forum, where trends in market entry and partnerships were highlighted. In China, cultivating guanxi (relationship-based business culture) is essential, cross-cultural advisory services can provide coaching on negotiation etiquette and long-term relationship-building to foster trust.
- New Market Openings
- Reduced competition: With EU and Chinese firms restricted, UK companies face fewer rivals in public tenders. Data from EU Tenders Electronic Daily shows a rise in non-EU bids for medical equipment in 2024, including from UK suppliers.
- Agile Sourcing: UK firms can offer compliant, flexible supply chains that adapt to evolving regulations.
- Rising Demand: Healthcare providers in China and the EU are diversifying suppliers amid volatility, with Deloitte’s global health care outlook emphasising supply chain resilience and diversification strategies. Surveys indicate that many EU hospitals plan to broaden sourcing by 2026 to mitigate risks.
SMEs, in particular, can seize these opportunities with targeted support to address resource limitations.
- Global Reputation for Excellence
UK medical device firms are renowned for:
-
- Regulatory integrity: Adherence to the highest compliance standards
- Innovative R&D: Pioneering Solutions that address emerging healthcare needs
- Quality and Safety: Consistent delivery of products that meet or exceed international benchmarks
While facing competition from non-EU players, swift strategic action—such as through industry reports like the Global MedTech Outlook—can amplify these strengths.
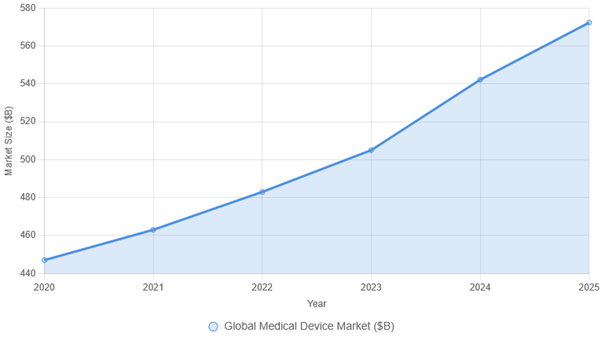
Opportunities Visualised: Growth of the Global Medical Device Market
Global market projections shown in USD. UK-specific figures in article are presented in GBP for policy relevance
How UK Businesses Can Succeed
UK firms, particularly SMEs, can thrive by adapting proactive strategies and seeking expert guidance.
Practical Steps for Market Entry
- Market Access: Monitor tenders in China and the EU with regulatory insights for competitive bidding. The UK Department for Business and Trade supported SMEs in 2024 EU market entries by navigating compliance, as seen in case studies of device experts.
- Partnership Building: Forge ties with local distributors, utilising cultural expertise for China’s guanxi or EU procurement. Cross-cultural advisory can include training on negotiation and relationship strategies.
- Compliance & Quality: Secure certifications and conduct audits to uphold trust amid stringent rules.
- Innovation & Strategy: Develop market-specific solutions, backed by cross-border R&D and intelligence on trends.
Risks such as supply chain costs or geopolitical shifts, necessitate robust planning. SMEs can mitigate these through cost-effective external support, avoiding the need for expansive in-house teams.
A Voice for Pragmatism and Partnership
UK businesses can advocate for collaboration, enhancing the UK’s image as a forward-thinking leader in global healthcare. By establishing reliable partnerships, they can achieve lasting market presence.
From Challenge to Catalyst
Trade tensions present an invitation to innovate and lead. With informed strategies, UK firms can drive growth in the medical device economy. For tailored market entry insights, consider contacting Excellence First Enterprise Consultancy (EFEC) for a complimentary initial consultation.
References:
- China Ministry of Commerce. (2025). Reciprocal Measures on EU Medical Devices.
- Deloitte Healthcare Report. (2025). EU Hospital Sourcing Trends.
- EU Tenders Electronic Daily. (2024). Medical Device Tender Data.
- European Commission. (2024). Public Procurement Restrictions on Medical Devices.
- Global MedTech Outlook. (2025). Competitive Landscape in Medical Devices.
- Statista. (2024). Global Medical Device Market Projections.
- UK Department for Business and Trade. (2025). UK SME Market Entry Support.
- UK MedTech Association. (2024). Case Study: UK Firm’s China Contract.